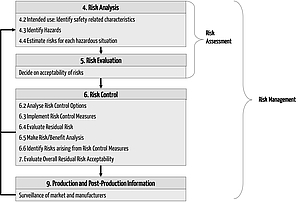
This is where some of the actual risk management takes place.
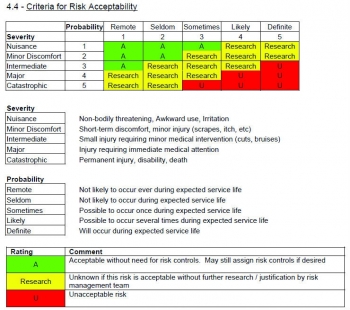
Visualizing your risk overview on a matrix diagram is a good idea:Īfter determining the level of acceptable risk, you’ll be able to clearly identify on the matrix if taking risk actions will be necessary. An unlikely, but possibly lethal or otherwise tragic hazardous situation will need to be treated as a high-risk situation. For instance, you will not have to worry too much about a hazardous situation that is very likely to occur, but will not cause much harm.
RISK ANALYSIS ISO 14971 TEMPLATE SERIES
You will need to identify the foreseeable series of events resulting from a hazard, and assess the risks based on this.ĭefining the values of severity and occurrence (probability) will help quantify and evaluate risks. All potential sources of harm (“hazards” in ISO 14971 terminology) will need to be analyzed to understand not only the causes, but also the risks these hazardous situations. By specifying the cope of your product and detailing its intended use, it will be easier for you to overview all the hazards that might be relevant, which is the second step of risk analysis. The risk analysis stage starts with defining the intended use of your product, as this will help guide your risk management efforts going forward. You will also need to make sure that your risk management plan & lifecycle is adequately documented: a risk management file needs to be established to contain all the documents and records that the risk management process produces. Essentially, what this means is that you will have to define the process you’ll be using, and the roles and responsibilities that will help you get there.

RISK ANALYSIS ISO 14971 TEMPLATE SOFTWARE
With or without the help of advanced software tools, you’ll have to follow certain steps in order to implement a thorough risk management lifecycle: 1) Establish risk management frameworkĪny risk management process that aims to be compliant with ISO 14971 and other regulations needs to start with establishing a risk management framework. This all-inclusive template may be used out of the box to reduce the time, effort, and cost requirements of compliance, and to make sure your risk management is as thorough as it needs to be. Implementing management policies, procedures and practices for managing risks is best supported by Intland’s Medical IEC 62304 and ISO 14971 Template 3.0. In the following, we’ll give a quick rundown of managing risks according to ISO 14971, an immensely complex topic on its own. Regardless of the large number of companies affected, interpreting the requirements of the various regulations, and putting into practice a system of risk management that fulfils all those requirements is still a challenge. So it’s clear that managing risks is a crucial process for all developers of medical devices and some other digital healthcare products. By FDA regulations, risk assessment is required as part of design validation (820.30 (g)). But other standards also require developers to implement risk management practices: ISO 13485, the regulation covering Quality Management Systems for medical devices, calls for the application of adequate “risk management throughout product realization”. One aspect of making sure that everything works as intended is, quite simply, to reduce or mitigate the chances of anything going wrong a crude definition of risk management itself.Īs mentioned above, the standard ISO 14971 focuses specifically on the application of risk management to medical devices. Standards regulating risk management in medical device developmentĭue to their safety-critical nature, developers of medical devices have to ensure that their products function reliably and as expected, causing no harm to patients, the operators of the device, or the environment. Developers of these products now face the challenge of having to adhere to continuously updated regulations in order to achieve compliance, and thus to be able to market their products.Īs one of the most critical requirements of several medical standards (and specifically covered by ISO 14971), the adequate management of product risks is becoming a source of headache for medical device developers around the world.

The group of products regulated by various medical standards is expanding to include everything from health monitoring smartphone apps to high-tech neurostimulation devices. That is, in our everyday lives, since digital healthcare technology is used even before (and specifically to prevent that) we become patients. With integrated, value-based healthcare moving health awareness and care “upstream” to prevent hospital care, medical devices are becoming more and more integrated in the lives of patients.
